|
In other words, even though an element group might break a trend, the elements within the group display periodic properties. Show how alloying introduces a defect into the metal, and how that makes the metal stronger. Alloys are often harder than metals composed of a pure element. However, the behavior of the noble gases is periodic. Steel is an alloy of iron with any of a number of other elements, such as chromium or vanadium. The alkali metals, also known as the alkali metal family is a group of six elements characterized by common physical and chemical properties, a similar electron configuration, and shared periodic trends. The noble gases are an exception to the trend since these elements have filled electron valence shells and electron affinity values approaching zero. Nonmetals usually have higher electron affinities than metals. Electron affinity increases moving across a period and decreases moving down a group. Electron Affinity - This is a measure of readily an atom accepts an electron.Atom and ion sizes shrink moving across a period because the increasing positive charge of the nucleus pulls in the electron shell. Although it might seem like increasing the number of protons and electrons in an atom would always increase its size, the atom size doesn't increase until a new electron shell is added. 
Ionic radius is the distance for ions of the atoms and follows the same trend. AP Chemistry: The Periodic Table of Elements. Atomic radius decreases moving left to right across a period and increases moving down a group. Atomic Radius - This is half the distance between the middle of two atoms just touching each other.Elements to the left are metals and nonmetals are to the right. The metalloid group separates the metals from the nonmetals. This periodic table shows the three different groups of elements. Electronegativity - A measure of how readily an atom forms a chemical bond. Electronegativity increases moving left to right across a period and decrease moving down a group. The elements of the periodic table can be broken into three different groups: Metals, Metalloids, and Nonmetals. 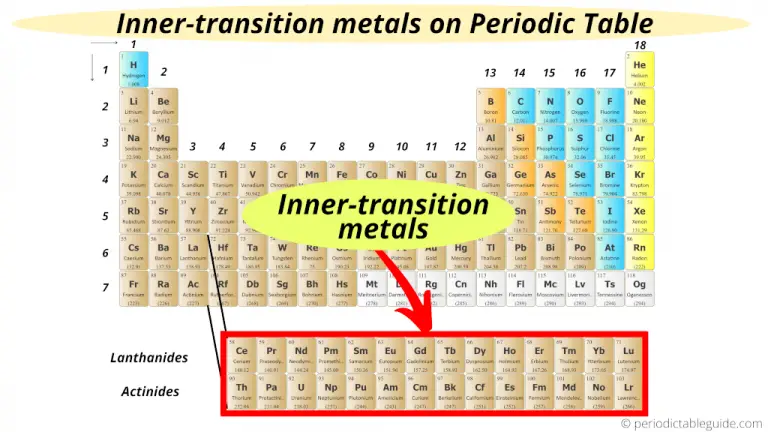
Ionization energy increases moving left to right across the table and decreases moving down a group. 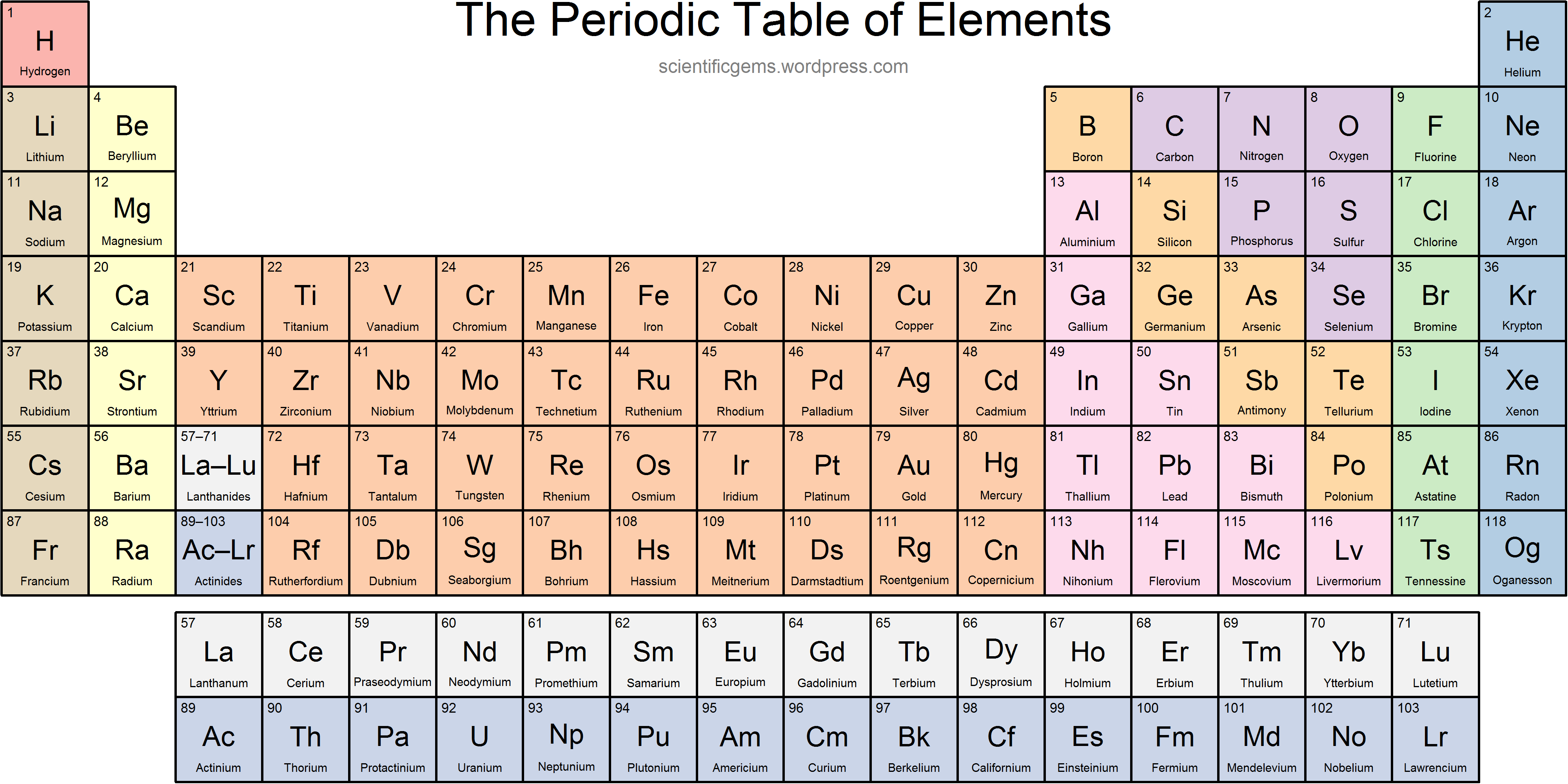
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
